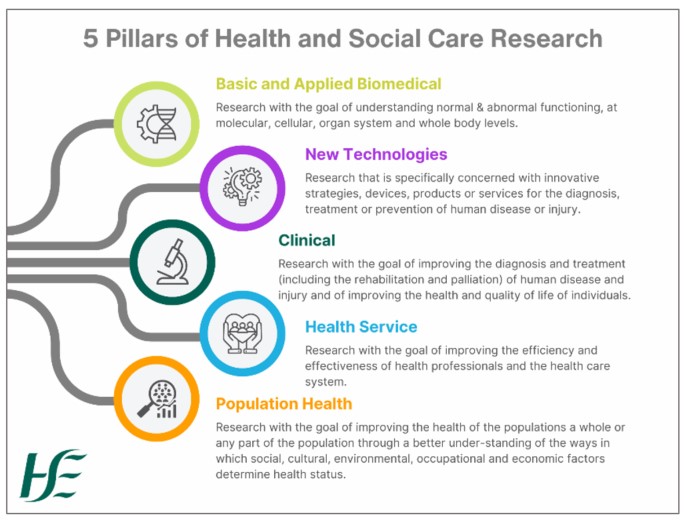
We refer to Health and Social Care Research as the attempt to derive generalisable or transferrable new knowledge to answer or refine relevant questions with scientifically sound methods in the areas of health and social care which includes basic and applied biomedical research, new technologies, clinical, health services and population health research. It refers to all health and social care research that requires the recruitment of participants via the HSE or HSE funded services or the use of their data or biomaterial, regardless of whether the research is led by the HSE or an external organisation.
A key feature of research, which differentiates it from other evidence generating activities, is that it is intentionally planned and designed using documented methodology which will allow results to be extrapolated or applied from the study sample to a larger population. This extrapolation / application is what the terms ‘generalisable’ and ‘transferable’ refer to. In the case of quantitative research, statistical methods are used to achieve results that are ‘generalisable’ from a sample to the sampled population. In the case of qualitative research, the context and findings are described and defined so that the conclusions can be applied or transferred to other settings.
When is Research Within the Scope of the RGMS Framework?
A study falls within the scope of the RGMS Framework if it involves the publicly funded health and social care service in specific ways. It must sit within one or more of the five research pillars and meet at least one of the following criteria:
- requires recruitment of participants via the publicly funded health and social care service
- requires the processing of personal data from service users’ healthcare records for research purposes
- requires the use of biological samples from service users for research purposes
- involves health and social care staff in the publicly funded health and social care service;
i. as participants.
ii. as researchers, when that research falls within the RGMS Scope. - is hosted within the publicly funded health and social care service and/or makes use of health and social care services infrastructure or requires support by healthcare services staff.
This also applies to health research taking place in external institutions such as third-level collaborative institutions and/or clinical research facilities (CRFs) when the research activities involve any of the aforementioned factors.
For further information please download:
Activities within the Scope of the HSE National Framework for the Governance, Management and Support of Research V2.0 April 2026 PDF 969KB
What is Not in Scope?
Some activities, even if methodologically rigorous or related to health services, fall outside the RGMS Framework. These include:
- clinical audits,
- standard service evaluation and development,
- health surveillance,
- normal and statutory public health work,
- advanced health analytics carried out by the service for the purpose of service planning or quality improvement.
Even when a study is out of scope, it may still require ethical oversight or governance approval at local level. For more information on how to differentiate research from other processes please see the National Centre for Clinical Audit – Nomenclature – A Glossary of Terms for Clinical Audit 2025
Examples
| Scenario | In Scope? | Rationale |
|---|---|---|
| A consultant in a public hospital establishes a registry to track patients with Polycystic Kidney Disease for the purposes of clinical audit and evaluation. | X Out of Scope | The primary purpose is audit and service evaluation, not research. |
| A university advertises a research study recruiting frail older adults using HSE home support for an intervention. | ✓ In Scope | Participants are identified via their use of HSE-funded services. |
| Clinical swabs are taken from gingivitis patients at HSE dental clinics are used to grow bacteria. No human tissue or patient data is used. | X Out of Scope | The research uses only microorganisms, not human samples or data. |
| Samples are collected from patients with Crohn’s disease during surgery to study gut flora gene expression and its link to patient outcomes. Clinical history is accessed. | ✓ In Scope | Access to patient clinical data and samples brings the study under the RGMS Framework. |
| A HSE physiotherapist recruits HSE patients in a Clinical Research Facility for a Master’s study. | ✓ In Scope | Involves HSE patients and infrastructure |
| An academic physiotherapist recruits HSE patients at a Clinical Research Facility for an observational study. | ✓ In Scope | HSE patients are involved; recruitment occurs through HSE- infrastructure. |
| A HSE community nurse, as part of their role, conducts a public perception survey of the home-help service via social media. | ✓ In Scope | The study is directly related to the nurse’s HSE role and context. |
| A HSE community nurse conducts personal research on ageing in her own time, recruiting via social media. | X Out of Scope | The project is not linked to the HSE role or infrastructure. |
| A researcher uses social media to recruit any nurses in Ireland to study the impact of night shifts. | X Out of Scope | Participants are not specifically recruited via HSE or funded services. |
| A researcher uses social media to recruit nurses working in HSE and Section 38 hospitals for a study on wellbeing. | ✓ In Scope | Targets staff based on their role within HSE-funded services. |
Why Does Scope Matter?
If your research is within the scope of the RGMS Framework, it must receive
- approval from a HSE or Section 38/39 Research Ethics Committee (or National Research Ethics Committee)
and - Institutional approval from the host site Research Office or equivalent
The study must also comply with HSE research policies.
Evidence generating activities that are out of scope may still require local governance, and researchers should confirm requirements with their senior management team before proceeding.
