The implementation of the HSE RGMS Framework requires not only the development of best practice approaches for research governance, but also the harmonisation and standardisation of those approaches across the system. This is essential to facilitate research which takes place in multiple sites, such as multisite clinical trials and national studies.
National Research and Development are working with a variety of key stakeholders to ensure a cohesive and coordinated approach to research governance is established in the health service:
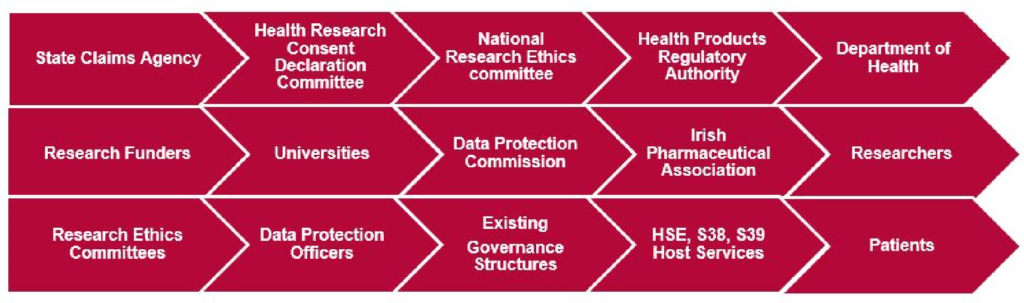
Work to date in this regard is taking place along multiple fronts:
- Harmonisation of protocols for data protection compliance in research
- Standard Contractual framework for research activity
- Standard Code of practice for HSE Research management
- Standard Code of Practice for HSE Reference Research Ethics committees
- National consent for research policy
Data protection compliance in research
HSE services have developed their own individual approaches for their research activity to comply with GDPR and the Health Research Regulations 2018. This has resulted in a very complex and fragmented compliance landscape, which makes collaborative and multisite research challenging.
The HSE National Research and Development team is working with key stakeholders to harmonise the approach to data protection governance in research. The aim is to develop agreed processes, tools, and guides to facilitate health research while keeping patient privacy rights at the forefront of that research. This include the development of:
- National standard Data Protection Impact Assessment (DPIA) for Research template for the health services together with a comprehensive user guide.
- A working framework to harmonise multisite DPIA reviews in the HSE and hospitals.
- The roll out of an international research data transfer risk assessment tool, to assist with the transfer of personal data for research purposes outside of Ireland/EU.
- A continuous training and development programme for research managers and other key staff is taken place in a number of pilot sites.
Legal Contract Framework
Research often requires the execution of collaborative legal contracts between a number of distinct groups/organisations (for example between collaborators, between sponsors and hospitals, etc). The National R&D team is working with key stakeholders to develop an agreement on a contractual legal framework for different types of research engagement (i.e clinical trials, collaborative projects, research commissions, etc). The objective of this framework is to:
- Reduce contract negotiation time and speed up study start up.
- Reduce legal costs.
- Standardisation of research contractual approach nationally within the health service.
The development of the suit of legal agreements for clinical trials commenced in 2022 in collaboration with the Irish Pharmaceutical healthcare Association (IPHA) with the launch of the template legal agreement for clinical trials involving one commercial sponsor and one hospital. The process is ongoing and the remaining clinical trials contract templates should be completed in 2023.
National HSE R&D is also collaborating with the Irish University sector to develop and agree a suit of contract templates for academically sponsored clinical trials and other templates that are commonly required for engagement with the academic community in health and social care research.
To support both hospitals and community organisations in both the use of the contract templates, and in the contracting process. National R&D is developing a Research Contract Framework Manual that will be launched in early 2024.
Standard Code of practice for HSE Research management
The HSE Health Region Research Office will carry out its activities in accordance to a standard code of practice which is currently being developed by National R&D in collaboration with the RGMS Framework implementation working group, with a expected completion date of end of 2023 or early 2024.
Standards for Research Ethics Committees
The reform of the HSE research ethics committee landscape will result in the establishment of Six HSE Reference RECS, all of which will work in accordance to a standard code of management and governance, with agreed standardised procedures to manage their operations in accordance to best practice.
In addition, minimum operational standards have also been developed for Hospital RECs.
To support HSE Reference RECs in operating to the standard code of practice and hospital RECS with the implementation of the minimum standards, the HSE will set up a REC support and co-ordination office as part of National R&D in 2024
National Consent for Research Policy
The HSE National Policy for Consent in Health and Social Care Research was launched by HSE National R&D in December 2024 and it is an invaluable resource for researchers, research ethics and patients alike to understand the requirements for consent for research in the HSE.
